
However the main downside is they are usually sensitive to electrolytes and sometimes shear. 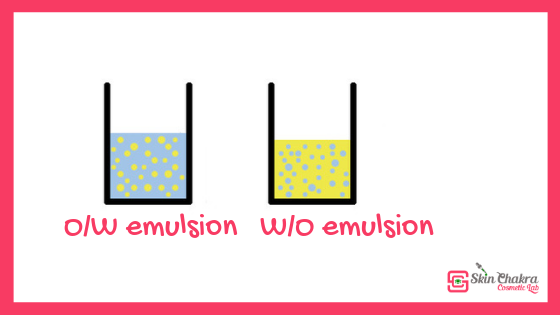
They have a lower usage level than traditional emulsifiers. They have a number of advantages, they are cold processable and so save energy and are quicker to process. If used as sole emulsifier without waxy ingredients they create gel-creams, serums. They are used alone or in combination with traditional emulsifiers. The lipophilic portion of the polymer adsorbs at the oil-water interface, and the hydrophilic portion swells in the water forming a gel network around oil droplets. This kind of emulsifier operates differently from the others. Examples: sepigel 305, sepiplus 400, simulgel, cosmedia ACE, viscoptima SE. There is a wide range available and they are generally more stable to electrolytes. They are versatile and because they do not have an overall charge and so are compatible with other emulsifier types. – Non-ionic emulsifiers (W/O or O/W) are the most common emulsifier type. They can be easier to preserve however some types, for example, the shorter chain monoalkyl analogues may cause irritation. These emulsifiers tend to be used in conditioners and give a silky, velvet, powdery, dry feel. – Cationic (O/W) emulsifiers are where the polar hydrophilic head groups carry a positive charge, such as behentrimonium methosulfate. These types of emulsifiers are however quite old fashioned, not as versatile, some may need a higher pH but on the plus side they tend to be quite cheap. 
Examples: sodium stearate, sodium stearyl glutamate, glyceryl stearate SE. – Anionic (O/W), this is where the hydrophilic head groups carry a negative charge. (In a w/o emulsion, the emulsifier’s orientation is reversed.) In this way, emulsifiers lower the interfacial tension between the oil and water phases, and form a protective layer around the oil phase keeping the oil droplets evenly dispersed and preventing them from flocculating.Ĭetyl alcohol and beeswax are not emulsifiers! When added to an o/w emulsion, the emulsifier surrounds the oil droplet with it’s oil-loving tail extending into the oil, and it’s water-loving head facing the water. Most emulsions we make are o/w which are generally, more stable and easier to formulate, and this is the type of emulsion we will be discussing in this article.Įmulsifiers contain both a water-loving head group and an oil-loving tail. The emulsifier we choose determines whether the emulsion will be o/w or w/o. 
Simple emulsions are either oil suspended in a water phase (o/w), or water suspended in oil (w/o). As an emulsion will eventually separate over time, our aim is delay this separation for as long as possible. However this is not a natural state – J W Gibbs, On the equilibrium of heterogeneous substances (1878), stated that “the only point in time where an emulsion is stable, is when it is completely separated”. Emulsions allow otherwise incompatible ingredients, such as oils, glycerin, actives, to be brought together in the same product.Īn emulsion can be defined as a dispersion of one material (usually the oil phase) inside another non-miscible (water) phase. Emulsions allow oils and butters to be applied to the skin in an aesthetically pleasing fashion and offers the formulator a great degree of formulation flexibility and easy modification of characteristics such as viscosity, feel and appearance. This article examines emulsions and provides guidance on how to create a stable product.Ī common complaint of non-water containing products, such as body butters and balms are they are greasy, heavy and can be grainy. The other (click here) you can use if you already have a recipe.Ĭreating an emulsion which is stable during the shelf life of the product is a challenge for most formulators. One also creates a formula for your specific skin and body type and includes stable emulsifiers and calculates the emulsifier level for you – (click here). We have two free calculators which both convert percentages to weight and work out the costs of the ingredients in your formula. As oil and water do not mix we add ingredients such as emulsifiers, stabilisers and use processing methods to help our lotion stay emulsified. Anyone who has made vinaigrette knows that oils do not dissolve in water no matter how long you shake them. Lotions contain a water phase and an oil phase.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
